Which of the Following Nuclei Would Be the Least Stable
Correct option is B 4. The stable elements at the end of the decay series all have a magic number of neutrons or protons.

Nuclear Chain Nuclear Physics Teaching Chemistry Science Chemistry
The unstable nuclei in order to become stable nuclei emit particles andor electromagnetic radiation.

. The correct order of True False in above statements is. BEmission of α particle is normally followed by emission of γ rays. A Z N Number Even Even Even.
The following table gives the distribution of the stable nuclei with respect to evenodd combinations of Z and N. When a nucleus in an excited state decays to the ground state the particle emitted is. Be 10 nuclei is unstable due to high np ratio which is 410.
The correct answer would be option D. What nuclei would be the least stable. B The light nuclei with A 20 are least stable.
Which statement is true regarding the stability of these nuclei. Nuclei with certain numbers of nucleons known as magic numbers are stable against nuclear decay. Nuclides containing odd numbers of both protons and neutrons are the least stable and this means more radioactive.
Rank the following isotopes in order of their radioactivity from the most radioactive to the least radioactive. The nuclei He-4 O-16 and Pb-208 82 protons and 126 neutrons that contain magic numbers of both neutrons and protons are particularly stable. ANuclear fission is normally followed by emission of β particles.
Assume they all have stable nuclei. Consider the following statements. Heavy nuclei with an even number of protons and an even number of neutrons are due to Pauli exclusion principle very stable thanks to the occurrence of paired spin.
On the other hand nuclei with an odd number of protons and neutrons are mostly unstable. C actinium-225 half-life 10 days. ANucleus D is the most stable and nucleus A is the least stable.
10 protons 12 neutrons because the closer the neutrons to the number of protons the more stable. Nuclides containing even numbers of both protons and neutrons are most stable and this. The three type of emission that originate from the nucleus are.
The isotopes with intermediate mass numbers 40 to 100 are most stable. The average binding energy for most of the nuclei is in the vicinity of 8 MeV. Lighter nuclei have np ratio around 1.
These comprise the first 82 elements from hydrogen to lead with the two exceptions technetium element 43 and promethium element 61 that do not have any stable nuclides. If a nuclei has even numbers of nucleons its generally more stable. DNuclei A and B are stable but nucleus B is more stable than nucleus A.
BNucleus C is stable whereas nuclei A B and D are not. Iron has the maximum average binding energy 879 MeV and thus its nucleus is thermodynamically most stable. The mass of an atom of carbon-12 Z 6 is less than the mass of 6 protons and 6 neutrons because of.
B uranium-238 half-life 45 billion years. C The curve has certain peaks indicating that certain nuclei like C He 12 6 4 2 and O 16 8 are much more stable than the nuclei in their vicinity. For example helium-4 is among the most abundant and stable nuclei in the universe.
CAs the mass number A increases the binding energy per nucleon in a nucleus increases. The time interval during which a nucleus has a 50 probability of decaying is its. These types of nuclei are said to be radioactive and the emission is called radioactivity.
These numbers of protons or neutrons 2 8 20. A nickel-59 half-life 75000 years. See the related Wikipedia linkLarger atomic nuclei.
Nuclei having binding energy per nucleon very near to 8 MeV are more or less stable. Of the known chemical elements 80 elements have at least one stable nuclide. It shows that an even-even combination of Z and N gives high stability and odd-odd the least.
As you go to heavier nuclei like uranium for instance thenucleus gets less stable. Light nucleus predicted to be stable despite having two strange quarks. 1 proton 3 neutrons.
An artists impression of a bound nucleus containing three normal nucleons which have up and down quarks and a. Correct answer to the question Which of the following nuclei would be most stable. Alpha particles α Beta particles β Gamma particles γ.
CNucleus A is the most stable and nucleus D is not stable. Which of the following noble gases would behave the LEAST like an ideal gas. Nuclei with even numbers of protons neutrons or both are more likely to be stable see Table 1.
Nuclei with magic numbers usually tend to be more stable.
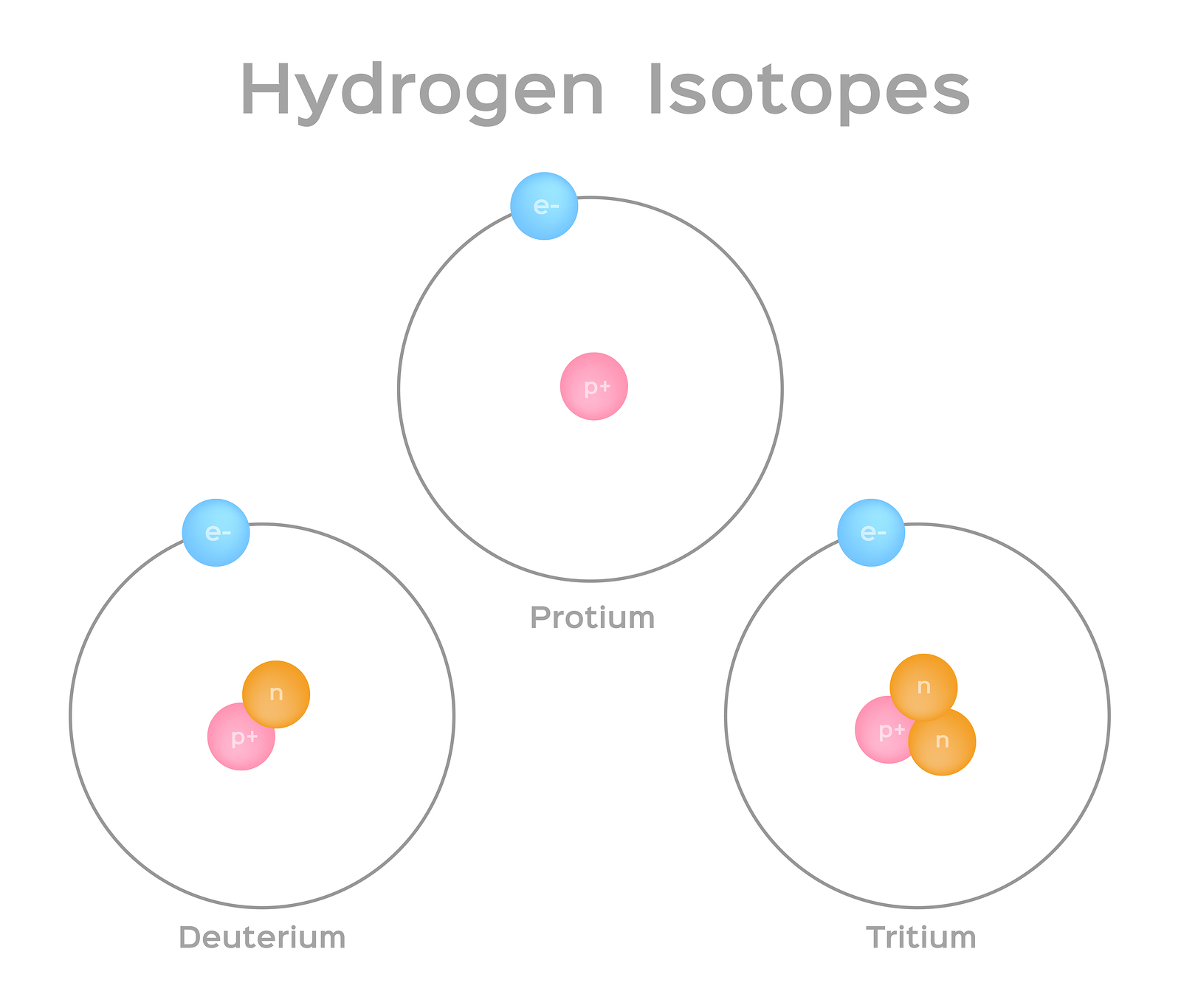
Isotope Basics Nidc National Isotope Development Center

Stable And Unstable Nuclei Radioactivity Physics Fuseschool Youtube

What Is Resonance Effect With Example Chemistry Notes Structural Formula Chemistry

Posts About Nuclear Physics On Reality Based World Nuclear Physics Physics Learn Physics

While It Was An Interesting Idea To Harness Fusion Power In The Earth S Own Core The Temperature Is Only 10 000 Kelvin Earth S Core Chemistry Projects Solving

Homework And Exercises Why Do We Have Minimum Deviation Angle In A Prism Physics Stack Exchange Physics And Mathematics Physics Weird Facts

Coulombic Force An Overview Sciencedirect Topics

The Seperation Of Forces And Evolution Of Matter Elementary Particle Astrophysics Physics

Traces Of Ancient Life Tell Story Of Early Diversity In Marine Ecosystems Study School Of Medicine Analysis

Stable And Unstable Isotopes Youtube

Stable And Unstable Nuclei Radioactivity Physics Fuseschool Youtube

Stable Nuclides An Overview Sciencedirect Topics

Unstable Jpg 773 368 Science Images Scientist Educational Projects

Tetryonics 56 07 Valence Electron Rules Dictate How Atoms Lose And Seek Electrons In Order To Fill Electron Orbitals In Sear Noble Gas Quantum Leap Electrons

Platinum On The Other Hand Has Pt190 78 Protons And Electron Pairs And At Least An Equal Number Of Neutrons Plus A Few To R Neutrons Protons Electrons
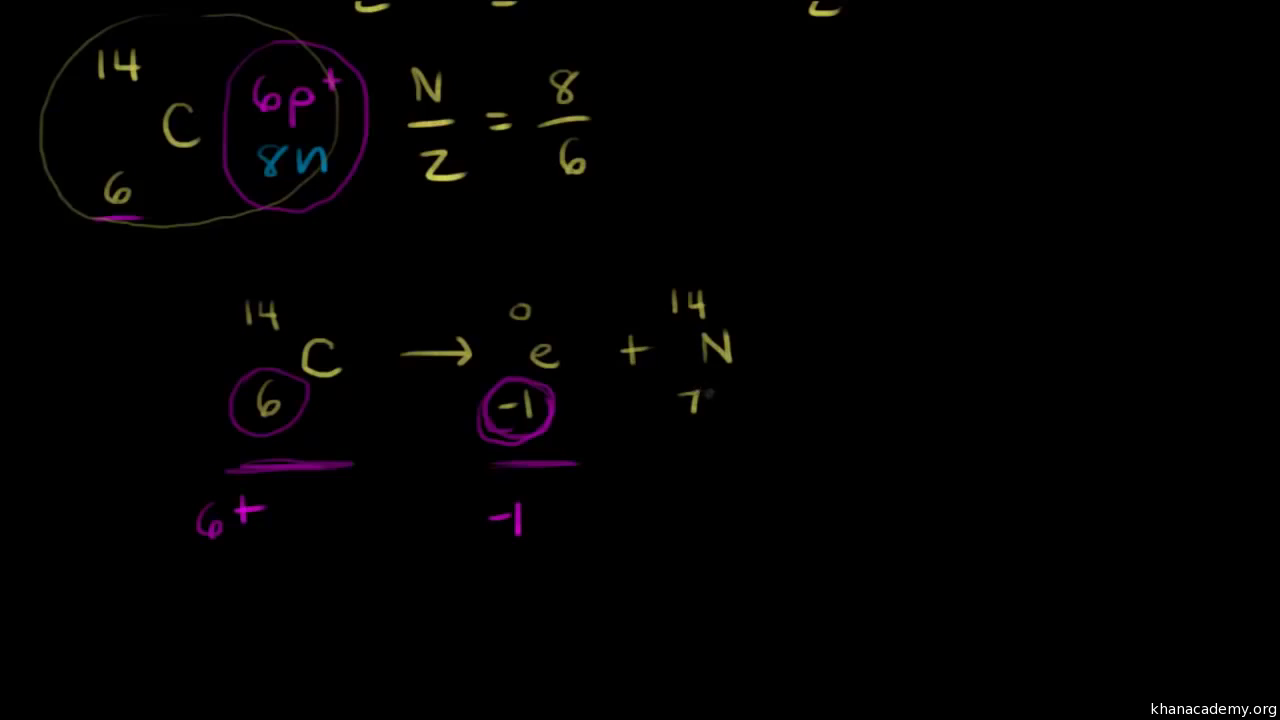
Nuclear Stability And Nuclear Equations Video Khan Academy

The Semi Empirical Mass Formula Binding Energy Deep Truths Physics

Medical Laboratory And Biomedical Science Csf Gram Stain Case Medical Laboratory Biomedical Science Stain
Comments
Post a Comment